Monomer: Styrene¶
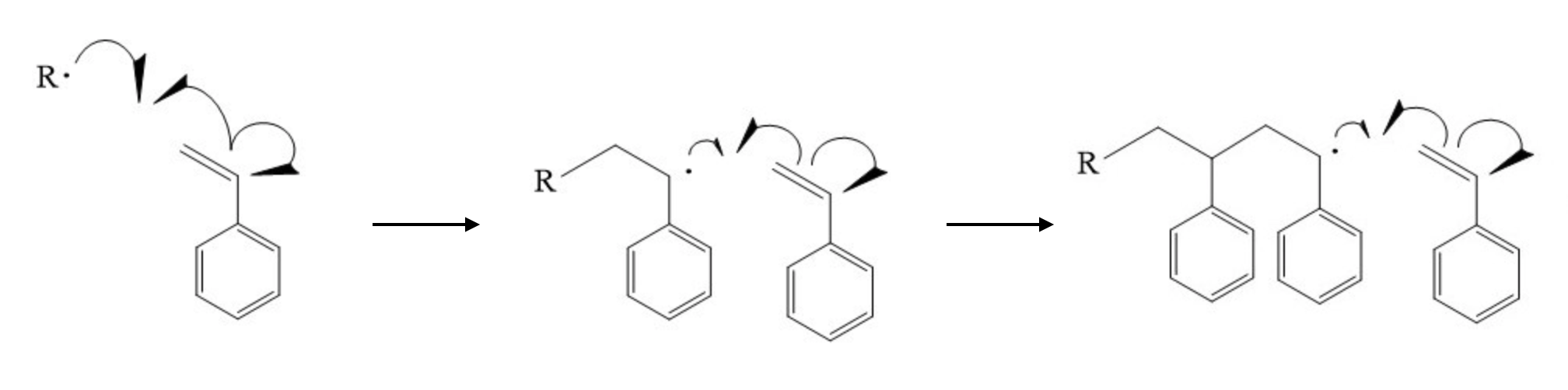
Fig. 6 Polymerization of styrene via carbon-carbon double-bond opening.¶
Styrene polymerizes by radical addition: the carbon-carbon double bond opens so that each radical carbon bonds to a radical carbon on an adjacent monomer (Fig. 6). htpolynet must be able to distinguish the two vinyl carbons, which we will call C1 (the internal, phenyl-bearing carbon) and C2 (the terminal methylene carbon).
Valence-conservation and the active form
As described in the user guide, htpolynet requires each reactive atom to carry a sacrificial hydrogen that is deleted when a new bond forms. The “inactive” form of styrene has a C=C double bond:
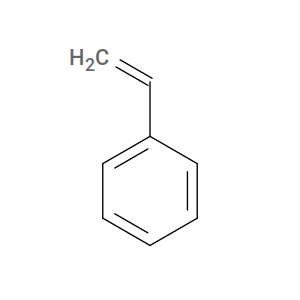
Fig. 7 Styrene (inactive form).¶
The active form we actually supply to htpolynet is ethylbenzene, which carries two extra hydrogens on what will become the reactive carbons:
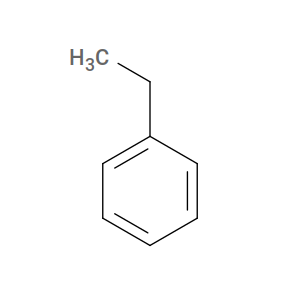
Fig. 8 Ethylbenzene (active form of styrene, monomer name STY).¶
Generating the mol2 file
The 1-polystyrene.sh script uses obabel and the SMILES string for ethylbenzene to generate a 3-D structure and rename the two reactive carbons:
$ obabel -:"C1=CC=CC=C1CC" -ismi -omol2 -h --gen3d --title styrene-active \
| sed s/" 7 C "/" 7 C1"/ \
| sed s/" 8 C "/" 8 C2"/ \
| sed s/"UNL1"/"STY "/ > lib/molecules/inputs/STY.mol2
The resulting file STY.mol2 looks like this:
@<TRIPOS>MOLECULE
STY
18 18 0 0 0
SMALL
GASTEIGER
@<TRIPOS>ATOM
1 C -0.7006 1.1795 -0.0272 C.ar 1 STY -0.0586
2 C 0.6928 1.2166 0.0538 C.ar 1 STY -0.0615
3 C 1.4223 0.0286 0.0810 C.ar 1 STY -0.0617
4 C 0.7581 -1.1951 0.0377 C.ar 1 STY -0.0615
5 C -0.6353 -1.2335 -0.0439 C.ar 1 STY -0.0586
6 C -1.3772 -0.0454 -0.0907 C.ar 1 STY -0.0476
7 C1 -2.8796 -0.0869 -0.2079 C.3 1 STY -0.0305
8 C2 -3.5705 -0.1623 1.1447 C.3 1 STY -0.0613
9 H -1.2556 2.1152 -0.0496 H 1 STY 0.0620
10 H 1.2096 2.1715 0.0912 H 1 STY 0.0618
11 H 2.5068 0.0578 0.1356 H 1 STY 0.0618
12 H 1.3256 -2.1221 0.0620 H 1 STY 0.0618
13 H -1.1387 -2.1963 -0.0798 H 1 STY 0.0620
14 H -3.1733 -0.9486 -0.8199 H 1 STY 0.0311
15 H -3.2298 0.7998 -0.7503 H 1 STY 0.0311
16 H -3.2680 -1.0573 1.6981 H 1 STY 0.0233
17 H -4.6567 -0.1988 1.0102 H 1 STY 0.0233
18 H -3.3353 0.7117 1.7611 H 1 STY 0.0233
@<TRIPOS>BOND
1 1 2 ar
2 2 3 ar
3 3 4 ar
4 4 5 ar
5 5 6 ar
6 1 6 ar
7 6 7 1
8 7 8 1
9 1 9 1
10 2 10 1
11 3 11 1
12 4 12 1
13 5 13 1
14 7 14 1
15 7 15 1
16 8 16 1
17 8 17 1
18 8 18 1
Atoms 7 and 8 (mol2 index) are now named C1 and C2 respectively. All other atoms keep generic names; only the atoms that htpolynet must track by name need to be uniquely labelled.
The next thing we consider is how to create the reaction dictionaries that describe the polymerization chemistry.